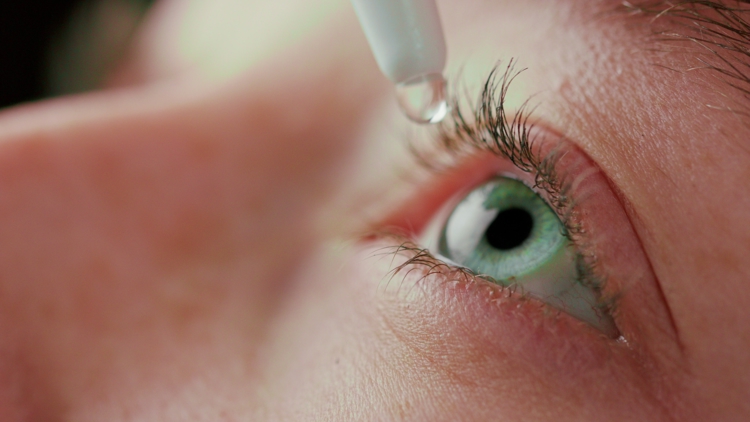
The products were sold at major retailers and may pose contamination risks, with consumers urged to stop use and check lot numbers.
Federal health officials are warning consumers to check their medicine cabinets after more than 3 million bottles of eye drops were recalled nationwide over potential safety concerns.
The U.S. Food and Drug Administration said K.C. Pharmaceuticals, Inc., based in Pomona, California, voluntarily recalled about 3.1 million bottles of various eye drop products due to a “lack of assurance of sterility.”
That means the company cannot confirm the products were manufactured in a way that prevents contamination from bacteria or other microbes.
The recall, which the FDA classified as Class II, indicates that use of the products could cause temporary or medically reversible health effects.
The affected products include a range of eye drops sold under multiple labels, such as artificial tears, advanced relief and redness relief formulas. They were distributed nationwide through major retailers and suppliers, including Walgreens, CVS, Kroger, H-E-B and other chains.
The FDA said most of the recalled products have expiration dates extending into 2026. While no specific contamination has been confirmed, officials said the lack of sterility assurance poses a potential risk to consumers.
Health officials are advising people to stop using the recalled eye drops immediately. Consumers should check product labels and lot numbers, particularly codes beginning with AC, AR, LT, SU, RG, RL, SY or AT with 2026 expiration dates.
Anyone who has used the products and experiences symptoms such as eye pain, redness or vision changes should seek medical attention.
The FDA said additional details, including specific product and lot information, are available in its enforcement report.
💌 Mantente al Día con lo Último del Entretenimiento Latino
Recibe noticias exclusivas de celebridades latinas, chismes virales, belleza, moda y entretenimiento — directo en tu correo.
Sin spam. Solo lo mejor de Atlanta Latinos Magazine.










